Revolutionary Discovery: IBM Builds the First "Half-Möbius" Molecule – A Premiere in Quantum Chemistry.
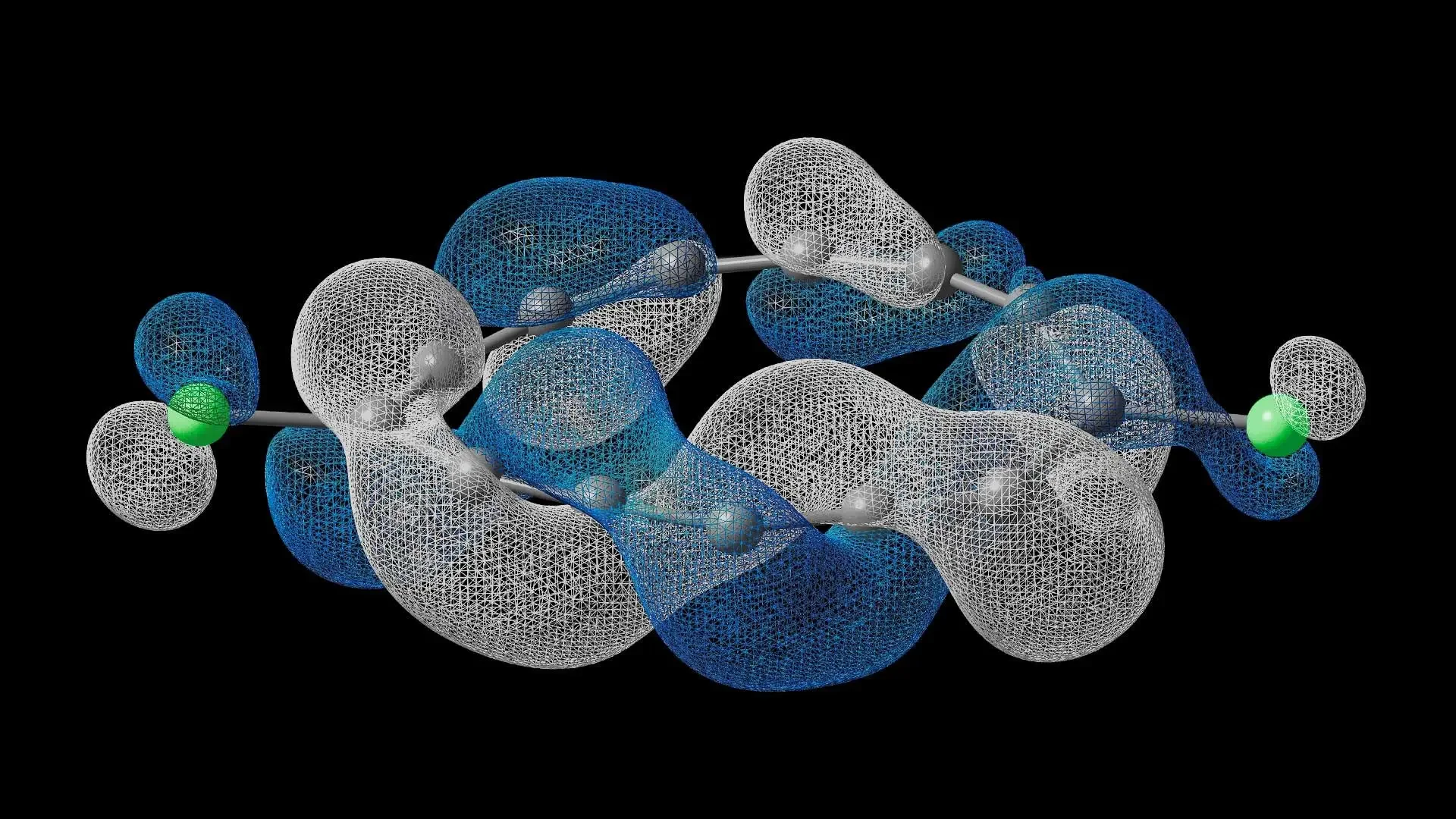
Dyson orbital for electron attachment, calculated using quantum hardware. Credit: IBM and the University of Manchester.
What's It About
The first molecule with half-Möbius electronic topology has been synthesized and characterized by an international team led by IBM Research, in collaboration with University of Manchester, Oxford, ETH Zurich, EPFL, and University of Regensburg. To the researchers' knowledge, a molecule with such topology has never been synthesized before, observed, or officially predicted.
The research was published in Science, where it details how it was assembled atom-by-atom using scanning probe microscopy to create a C₁₃Cl₂ molecule. To confirm the screw-like formation of the electrons, the team used an IBM Heron quantum processor to simulate it with high fidelity, surpassing the limits of classical computers.
How They Got the Results
Researchers at IBM built the molecule, with the chemical formula C₁₃Cl₂, atom by atom. They started from a special "template" created at the University of Oxford, then removed individual atoms one by one with precise electrical pulses – all in an extremely clean vacuum, at temperatures near absolute zero.
They used two "nanoscopic" microscopy techniques invented right at IBM: one measures electrical current (STM), the other atomic forces (AFM). By combining these images with simulations on a quantum computer, they discovered a completely new electronic structure: electrons "twist" 90 degrees with each lap around the molecule, returning to the initial state only after four full laps – like a half-twisted paper strip ("half-Möbius"), unlike any known molecule.
What's magic: this twist isn't accidental, but controllable – the molecule can reversibly switch between "right", "left", or "normal" states. We no longer "discover" electron properties; we build them intentionally, like an engineer designing a perfect screwdriver.
Future Implications
With these types of research, the need to increase computational resources grows, overwhelming classical computers. To solve this problem, we can turn to quantum computing.
Quantum computers are different by nature and operate under the same quantum mechanics laws that govern electrons in molecules, and they can represent these systems directly, not through approximation. This quality can offer capabilities with huge potential for quantum computers to contribute to real-world experiments, through the integration of quantum processing units (QPUs), CPUs, and GPUs.
Conclusion
And with this discovery, besides demonstrating the importance of quantum computers in quantum simulation, there's also the advance brought to chemistry, showing that electronic topology can be deliberately designed, not just found in nature.
What do you think? Comment!